- Thyroid
- Comparative Analysis of Driver Mutations and Transcriptomes in Papillary Thyroid Cancer by Region of Residence in South Korea
-
Jandee Lee, Seonhyang Jeong, Hwa Young Lee, Sunmi Park, Meesson Jeong, Young Suk Jo
-
Endocrinol Metab. 2023;38(6):720-729. Published online November 6, 2023
-
DOI: https://doi.org/10.3803/EnM.2023.1758
-
-
 Abstract Abstract
 PDF PDF Supplementary Material Supplementary Material PubReader PubReader  ePub ePub
- Background
Radiation exposure is a well-known risk factor for papillary thyroid cancer (PTC). South Korea has 24 nuclear reactors in operation; however, no molecular biological analysis has been performed on patients with PTC living near nuclear power plants.
Methods
We retrospectively included patients with PTC (n=512) divided into three groups according to their place of residence at the time of operation: inland areas (n=300), coastal areas far from nuclear power plants (n=134), and nuclear power plant areas (n=78). After propensity score matching (1:1:1) by age, sex, and surgical procedure, the frequency of representative driver mutations and gene expression profiles were compared (n=50 per group). Epithelial-mesenchymal transition (EMT), BRAF, thyroid differentiation, and radiation scores were calculated and compared.
Results
No significant difference was observed in clinicopathological characteristics, including radiation exposure history and the frequency of incidentally discovered thyroid cancer, among the three groups. BRAFV600E mutation was most frequently detected in the groups, with no difference among the three groups. Furthermore, gene expression profiles showed no statistically significant difference. EMT and BRAF scores were higher in our cohort than in cohorts from Chernobyl tissue bank and The Cancer Genome Atlas Thyroid Cancer; however, there was no difference according to the place of residence. Radiation scores were highest in the Chernobyl tissue bank but exhibited no difference according to the place of residence.
Conclusion
Differences in clinicopathological characteristics, frequency of representative driver mutations, and gene expression profiles were not observed according to patients’ region of residence in South Korea.
- Clinical Study
- Lactate Dehydrogenase A as a Potential New Biomarker for Thyroid Cancer
-
Eun Jeong Ban, Daham Kim, Jin Kyong Kim, Sang-Wook Kang, Jandee Lee, Jong Ju Jeong, Kee-Hyun Nam, Woong Youn Chung, Kunhong Kim
-
Endocrinol Metab. 2021;36(1):96-105. Published online February 24, 2021
-
DOI: https://doi.org/10.3803/EnM.2020.819
-
-
5,816
View
-
188
Download
-
14
Web of Science
-
13
Crossref
-
 Abstract Abstract
 PDF PDF Supplementary Material Supplementary Material PubReader PubReader  ePub ePub
- Background
Several cancers show increased levels of lactate dehydrogenase A (LDHA), which are associated with cancer progression. However, it remains unclear whether LDHA levels are associated with papillary thyroid cancer (PTC) aggressiveness or with the presence of the PTC prognostic marker, the BRAFV600E mutation. This study aimed to evaluate the potential of LDHA as a PTC prognostic marker.
Methods
LDHA expression was examined in 83 PTC tissue specimens by immunohistochemistry. Human thyroid cell lines were genetically manipulated to overexpress BRAFV600E or were treated with a BRAF-specific short hairpin RNA (shBRAF), whose effects on LDHA expression were evaluated by Western blotting. Data from 465 PTC patients were obtained from The Cancer Genome Atlas (TCGA) database and analyzed to validate the in vitro results.
Results
LDHA was aberrantly overexpressed in PTC. Intense immunostaining for LDHA was observed in PTC specimens carrying mutated BRAF, whereas the intensity was less in wild-type BRAF samples. Overexpression of BRAFV600E resulted in LDHA upregulation, whereas treatment with shBRAF downregulated LDHA in human thyroid cell lines. Furthermore, LDHA mRNA expression was significantly elevated and associated with BRAFV600E expression in thyroid cancer tissues from TCGA database. Additionally, LDHA overexpression was found to be correlated with aggressive clinical features of PTC, such as lymph node metastases and advanced tumor stages.
Conclusion
LDHA overexpression is associated with the BRAFV600E mutation and an aggressive PTC behavior. Therefore, LDHA may serve as a biomarker and therapeutic target in PTC.
-
Citations
Citations to this article as recorded by  - Integrated proteogenomic and metabolomic characterization of papillary thyroid cancer with different recurrence risks
Ning Qu, Di Chen, Ben Ma, Lijun Zhang, Qiuping Wang, Yuting Wang, Hongping Wang, Zhaoxian Ni, Wen Wang, Tian Liao, Jun Xiang, Yulong Wang, Shi Jin, Dixin Xue, Weili Wu, Yu Wang, Qinghai Ji, Hui He, Hai-long Piao, Rongliang Shi
Nature Communications.2024;[Epub] CrossRef - Peripheral lymphocytes and lactate dehydrogenase correlate with response and survival in head and neck cancers treated with immune checkpoint inhibitors
Cassie Pan, Qian Vicky Wu, Jenna Voutsinas, Jeffrey J. Houlton, Brittany Barber, Zain H. Rizvi, Emily Marchiano, Neal Futran, George E. Laramore, Jay J. Liao, Upendra Parvathaneni, Renato G. Martins, Jonathan R. Fromm, Cristina P. Rodriguez
Cancer Medicine.2023; 12(8): 9384. CrossRef - LncRNA GLTC targets LDHA for succinylation and enzymatic activity to promote progression and radioiodine resistance in papillary thyroid cancer
Liang Shi, Rui Duan, Zhenhua Sun, Qiong Jia, Wenyu Wu, Feng Wang, Jianjun Liu, Hao Zhang, Xue Xue
Cell Death & Differentiation.2023; 30(6): 1517. CrossRef - Integrated analysis of circulating and tissue proteomes reveals that fibronectin 1 is a potential biomarker in papillary thyroid cancer
Guochao Ye, Xiaomei Zhang, Mansheng Li, Zixiang Lin, Yongcan Xu, Haoru Dong, Jie Zhou, Jiaqi Zhang, Sheng Wang, Yunping Zhu, Xiaobo Yu, Xu Qian
BMC Cancer.2023;[Epub] CrossRef - Targeting metabolism by B-raf inhibitors and diclofenac restrains the viability of BRAF-mutated thyroid carcinomas with Hif-1α-mediated glycolytic phenotype
Marianna Aprile, Simona Cataldi, Caterina Perfetto, Antonio Federico, Alfredo Ciccodicola, Valerio Costa
British Journal of Cancer.2023; 129(2): 249. CrossRef - circNFATC3 facilitated the progression of oral squamous cell carcinoma via the miR-520h/LDHA axis
Hongguo Xie, Xiaopeng Lu
Open Medicine.2023;[Epub] CrossRef - The potential role of reprogrammed glucose metabolism: an emerging actionable codependent target in thyroid cancer
Sai-li Duan, Min Wu, Zhe-Jia Zhang, Shi Chang
Journal of Translational Medicine.2023;[Epub] CrossRef - CENPE and LDHA were potential prognostic biomarkers of chromophobe renal cell carcinoma
Hui-feng Wu, Hao Liu, Zhe-wei Zhang, Ji-min Chen
European Journal of Medical Research.2023;[Epub] CrossRef - Classification for Staging and Managing Patients with Biopolymer-induced Human Adjuvant Disease
Jaime Eduardo Pachón Suárez, Marcela C. Salazar, Victor Z. Rizo
Plastic and Reconstructive Surgery - Global Open.2022; 10(2): e4137. CrossRef - Development of Metabolic Synthetic Lethality and Its Implications for Thyroid Cancer
Sang-Hyeon Ju, Seong Eun Lee, Yea Eun Kang, Minho Shong
Endocrinology and Metabolism.2022; 37(1): 53. CrossRef - Drug delivery for metabolism targeted cancer immunotherapy
Taravat Khodaei, Sahil Inamdar, Abhirami P. Suresh, Abhinav P. Acharya
Advanced Drug Delivery Reviews.2022; 184: 114242. CrossRef - Sulfur quantum dot based fluorescence assay for lactate dehydrogenase activity detection
Shengnan Fan, Xiaoqing Li, Fanghui Ma, Minghui Yang, Juan Su, Xiang Chen
Journal of Photochemistry and Photobiology A: Chemistry.2022; 430: 113989. CrossRef - STAT3/LINC00671 axis regulates papillary thyroid tumor growth and metastasis via LDHA-mediated glycolysis
Nan Huo, Rui Cong, Zhi-jia Sun, Wen-chao Li, Xiang Zhu, Chun-yuan Xue, Zhao Chen, Lu-yuan Ma, Zhong Chu, Yu-chen Han, Xiao-feng Kang, Song-hao Jia, Nan Du, Lei Kang, Xiao-jie Xu
Cell Death & Disease.2021;[Epub] CrossRef
- Endocrine Research
- Liver X Receptor β Related to Tumor Progression and Ribosome Gene Expression in Papillary Thyroid Cancer
-
Seonhyang Jeong, In-Kyu Kim, Hyunji Kim, Moon Jung Choi, Jandee Lee, Young Suk Jo
-
Endocrinol Metab. 2020;35(3):656-668. Published online August 20, 2020
-
DOI: https://doi.org/10.3803/EnM.2020.667
-
-
6,605
View
-
132
Download
-
8
Web of Science
-
9
Crossref
-
 Abstract Abstract
 PDF PDF Supplementary Material Supplementary Material PubReader PubReader  ePub ePub
- Background
Intracellular lipid deposition has been reported in thyroid glands in obese animal and human. To understand the regulatory mechanism of lipid metabolism in thyroid cancer, we investigated the expression status of liver X receptor (LXR) and analyzed its clinicopathological characteristics and molecular biological features.
Methods
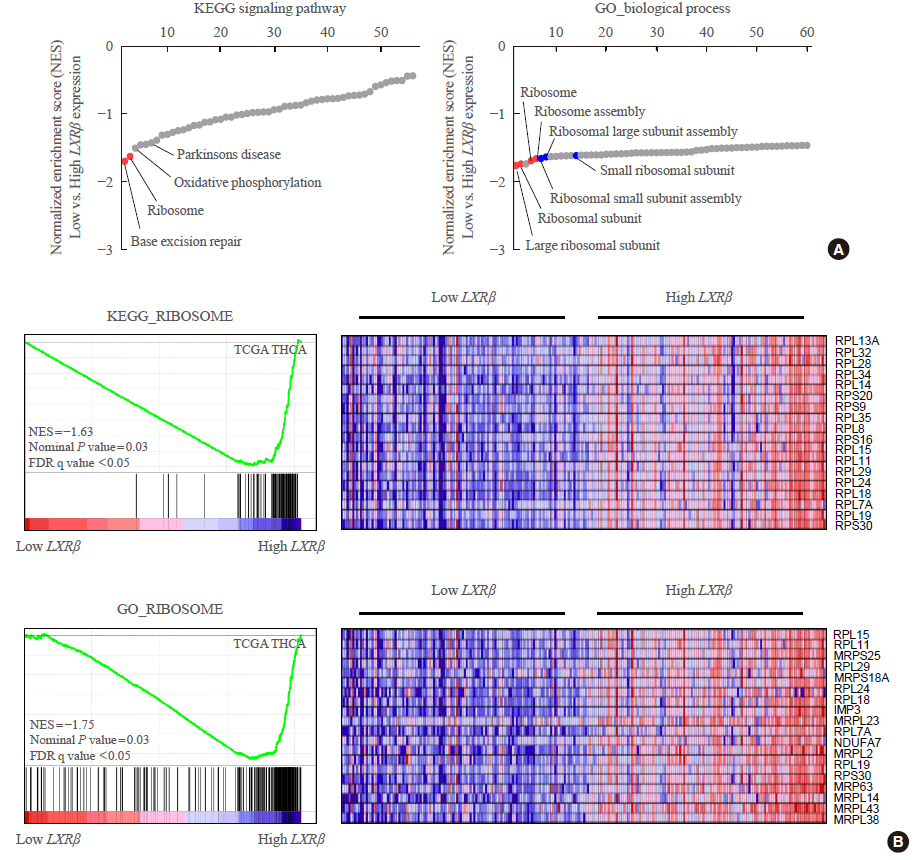
Expression status of LXR and its transcriptional targets in human cancers were analyzed using The Cancer Genome Atlas (TCGA). The gene-sets related to high LXRβ expression was investigated by gene set enrichment analysis (GSEA) using Kyoto Encyclopedia of Genes and Genomes (KEGG) signaling pathways and gene ontology biologic process. Quantitative reverse transcription polymerase chain reaction was performed in thyroid cancer samples using our validation cohort.
Results
In contrast to low expression of LXRα, LXRβ was highly expressed in thyroid cancer compared to the other types of human cancers. High LXRβ expression was correlated with the expression of LXRβ transcriptional targets genes, such as apolipoprotein C1 (APOC1), APOC2, apolipoprotein E (APOE), ATP binding cassette subfamily G member 8 (ABCG8), sterol regulatory elementbinding protein 1c (SREBP1c), and SPOT14. Furthermore, High LXRβ expression group indicated poor clinicopathological characteristics and aggressive molecular biological features independently from the drive mutation status. Mechanistically, high LXRβ expression was coordinately related to ribosome-related gene sets.
Conclusion
The mechanistic link between LXRβ and ribosomal activity will be addressed to develop new diagnostic and therapeutic targets in thyroid cancers.
-
Citations
Citations to this article as recorded by  - ApoC1 promotes glioma metastasis by enhancing epithelial-mesenchymal transition and activating the STAT3 pathway
Rui Liang, Guofeng Zhang, Wenhua Xu, Weibing Liu, Youjia Tang
Neurological Research.2023; 45(3): 268. CrossRef - The Novel RXR Agonist MSU-42011 Differentially Regulates Gene Expression in Mammary Tumors of MMTV-Neu Mice
Lyndsey A. Reich, Ana S. Leal, Edmund Ellsworth, Karen T. Liby
International Journal of Molecular Sciences.2023; 24(5): 4298. CrossRef - The Role of Apolipoproteins in the Commonest Cancers: A Review
Nour M. Darwish, Mooza Kh. Al-Hail, Youssef Mohamed, Rafif Al Saady, Sara Mohsen, Amna Zar, Layla Al-Mansoori, Shona Pedersen
Cancers.2023; 15(23): 5565. CrossRef - Apolipoproteins: New players in cancers
Yingcheng He, Jianrui Chen, Yanbing Ma, Hongping Chen
Frontiers in Pharmacology.2022;[Epub] CrossRef - Simultaneous Expression of Long Non-Coding RNA FAL1 and Extracellular Matrix Protein 1 Defines Tumour Behaviour in Young Patients with Papillary Thyroid Cancer
Seonhyang Jeong, Seul-Gi Lee, Hyunji Kim, Gibbeum Lee, Sunmi Park, In-Kyu Kim, Jandee Lee, Young-Suk Jo
Cancers.2021; 13(13): 3223. CrossRef - Using BioPAX-Parser (BiP) to enrich lists of genes or proteins with pathway data
Giuseppe Agapito, Mario Cannataro
BMC Bioinformatics.2021;[Epub] CrossRef - Comprehensive Analysis of Prognostic Alternative Splicing Signature Reveals Recurrence Predictor for Papillary Thyroid Cancer
Mian Liu, Rooh Afza Khushbu, Pei Chen, Hui-Yu Hu, Neng Tang, Deng-jie Ou-yang, Bo Wei, Ya-xin Zhao, Peng Huang, Shi Chang
Frontiers in Oncology.2021;[Epub] CrossRef - Metabolic Reprogramming of Thyroid Cancer Cells and Crosstalk in Their Microenvironment
Lisha Bao, Tong Xu, Xixuan Lu, Ping Huang, Zongfu Pan, Minghua Ge
Frontiers in Oncology.2021;[Epub] CrossRef - Cooperative Subtype Switch of Thyroid Hormone Receptor and Nuclear Receptor Corepressor Related Epithelial–Mesenchymal Transition in Papillary Thyroid Cancer
Seonhyang Jeong, Seul Gi Lee, Hyunji Kim, Gibbeum Lee, Sunmi Park, In-Kyu Kim, Jandee Lee, Young Suk Jo
International Journal of Thyroidology.2021; 14(2): 152. CrossRef
|